07 October 2021
A historic milestone in the global effort to combat malaria: the World Health Organization (WHO) has approved the first vaccine to address the disease. The vaccine, RTS,S , also known by its brand name Mosquirix, was developed by the British pharmaceutical giant GSK. To date, it has been administered to more than 800,000 children in Ghana, Kenya, and Malawi as part of a pilot immunization program that commenced in 2019.
The vaccine's efficacy is estimated at approximately 39% in preventing infection and 29% in preventing severe malaria cases resulting in hospitalization. A study conducted in the UK, which evaluated the combined use of the vaccine and antimalarial medications, found a 70% reduction in hospitalizations and fatalities. In 2019, malaria claimed 409,000 lives globally, the vast majority of which were in Africa, including over 270,000 children under the age of five. Meanwhile, the Jenner Institute at Oxford is developing another malaria vaccine with a claimed efficacy of approximately 77% (based on a trial conducted in Burkina Faso among 450 children).
About the Vaccine: The immunization schedule consists of three injections administered at one-month intervals, followed by a fourth booster dose 18 months after the third. It is indicated for children aged 6 weeks to 17 months (providing protection against both malaria and Hepatitis B).
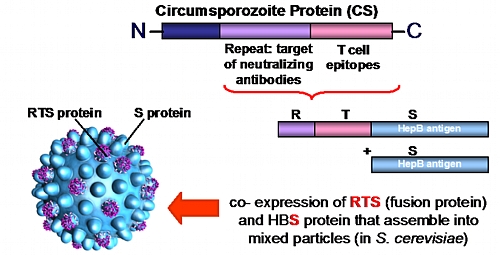
Technical Specifications: The vaccine is a recombinant (chimeric) vaccine containing the parasite’s Circumsporozoite protein (CSP). The CSP protein is characterized by extremely low variability, making it an ideal target for vaccine development. A segment of the CSP protein, which includes the repeat region (R) (containing neutralizing antibody epitopes) and T-cell epitopes, is fused with the Hepatitis B virus S-protein; together, they form the segment known as RTS.
This segment is then co-expressed with the Hepatitis B virus envelope protein (S) to form RTS,S. These proteins are produced using a yeast platform (Saccharomyces cerevisiae), where they spontaneously assemble into Virus-Like Particles (VLPs). When combined with an adjuvant (AS01), they constitute the final vaccine, which serves as a dual-target immunization for both Malaria and Hepatitis B.
In an article published in the BMJ in 2020, criticism was leveled at the ethical conduct of this pilot.
Doshi P. WHO’s malaria vaccine study represents a “serious breach of international ethical standards”. bmj. 2020 Feb 26;368.